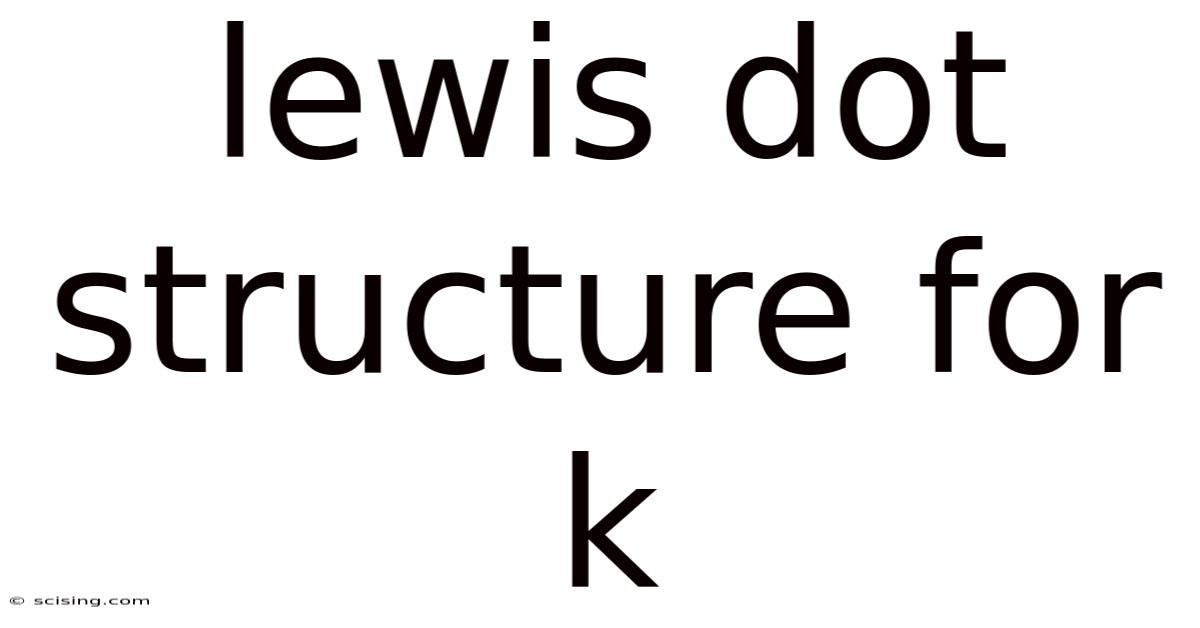Understanding Lewis Dot Structures: A Deep Dive into Potassium (K)
Lewis dot structures are a fundamental concept in chemistry, providing a simple yet powerful way to visualize the valence electrons of an atom and how they participate in chemical bonding. On the flip side, this article will delve deep into understanding Lewis dot structures, particularly focusing on potassium (K), a highly reactive alkali metal. That said, we'll explore its electronic configuration, how to draw its Lewis dot structure, and its implications in chemical reactions. Understanding potassium's Lewis dot structure is crucial for comprehending its behavior and its role in various chemical processes.
Introduction to Lewis Dot Structures
A Lewis dot structure, also known as an electron dot diagram, is a visual representation of the valence electrons of an atom. Valence electrons are the electrons located in the outermost shell of an atom, and they are the ones primarily involved in forming chemical bonds. Still, these structures are named after Gilbert N. Lewis, who introduced this notation in 1916.
The core principle behind Lewis dot structures is that each valence electron is represented by a dot placed around the element's symbol. As an example, hydrogen (H) with one valence electron is represented as H•, while oxygen (O) with six valence electrons is represented as :Ö:. The dots are strategically placed to represent the arrangement of electrons within the atom's outermost shell, and this arrangement dictates how the atom will bond with other atoms Most people skip this — try not to..
Understanding Lewis structures helps in predicting the type of bonds an atom will form (ionic, covalent, or metallic), its reactivity, and the shape of the resulting molecule. This makes it an indispensable tool for students and professionals alike in chemistry and related fields.
Electronic Configuration of Potassium (K)
Potassium (K), with an atomic number of 19, belongs to Group 1 (alkali metals) in the periodic table. Its electronic configuration is [Ar] 4s¹. Worth adding: the [Ar] represents the electronic configuration of Argon, a noble gas with a filled outermost shell (3s²3p⁶). Put another way, potassium has 19 electrons arranged in various energy levels or shells. The crucial part for our Lewis dot structure is the 4s¹ – meaning potassium has only one electron in its outermost shell (valence shell).
Drawing the Lewis Dot Structure for Potassium (K)
Because potassium has only one valence electron, drawing its Lewis dot structure is straightforward. Simply write the element symbol (K) and place one dot next to it Simple, but easy to overlook..
K•
This single dot represents the solitary valence electron in potassium's outermost shell. This single valence electron is readily lost to achieve a stable electron configuration similar to Argon (a noble gas with a full octet). This characteristic explains potassium's high reactivity.
Potassium's Reactivity and its Lewis Dot Structure
The Lewis dot structure for potassium clearly illustrates its high reactivity. The single valence electron is easily lost to form a +1 ion (K⁺), achieving a stable electron configuration identical to Argon. This process is exothermic, meaning it releases energy. The loss of the electron results in a positively charged ion because the number of protons (19) exceeds the number of electrons (18).
This tendency to lose an electron and form a positive ion is the defining characteristic of alkali metals and is a direct consequence of its electronic configuration and its Lewis dot structure. The driving force behind this behavior is the atom's quest to achieve a stable, low-energy configuration. Noble gases, with their filled outermost electron shells, are the epitome of this stability But it adds up..
Ionic Bonding and Potassium
Potassium's tendency to readily lose its valence electron makes it highly reactive and prone to forming ionic bonds. Ionic bonds form between atoms with significantly different electronegativities. Electronegativity is the measure of an atom's ability to attract electrons in a chemical bond. Because of that, potassium, being an alkali metal, has low electronegativity. It readily loses its electron to a more electronegative atom, such as chlorine (Cl), which has seven valence electrons and a high electronegativity.
When potassium reacts with chlorine, potassium loses its valence electron to chlorine. Potassium becomes a positively charged ion (K⁺), and chlorine becomes a negatively charged ion (Cl⁻). The electrostatic attraction between these oppositely charged ions forms an ionic bond, creating potassium chloride (KCl), a common salt.
Comparing Potassium's Reactivity with Other Alkali Metals
All alkali metals, like lithium (Li), sodium (Na), and rubidium (Rb), exhibit similar reactivity due to their single valence electron. The increasing atomic radius down the group (from Li to Rb) makes the outermost electron less tightly bound, thus enhancing reactivity. Because of that, potassium is more reactive than sodium and lithium because its valence electron is further away from the nucleus, making it easier to remove. Even so, their reactivity varies slightly. This trend is directly reflected in their Lewis dot structures – each has one dot representing the single valence electron, but the ease with which this electron is lost varies slightly.
Potassium in Biological Systems
Potassium plays a vital role in biological systems. The single positive charge and its ability to move across cell membranes are key to its biological functions. Its ionic form (K⁺) is crucial for maintaining proper electrolyte balance, nerve impulse transmission, and muscle contraction. The body carefully regulates potassium levels, as imbalances can have serious consequences But it adds up..
Applications of Potassium and its Compounds
Potassium and its compounds have numerous applications. Potassium nitrate (KNO₃) is a vital component of fertilizers. Potassium hydroxide (KOH) is used in the production of soaps and detergents. Potassium permanganate (KMnO₄) is a strong oxidizing agent used in various chemical processes and as a disinfectant. These applications directly relate to potassium's chemical properties, which are easily understood using its Lewis dot structure.
Frequently Asked Questions (FAQ)
Q: Why is the Lewis dot structure important for understanding potassium's reactivity?
A: The Lewis dot structure visually demonstrates potassium's single valence electron, readily lost to achieve a stable octet configuration. This clearly shows why potassium is highly reactive and readily forms ionic bonds Turns out it matters..
Q: Can potassium form covalent bonds?
A: While potassium predominantly forms ionic bonds, it's theoretically possible for potassium to participate in covalent bonding under highly specific and unusual conditions. Even so, this is extremely rare. Its overwhelming tendency is towards ionic bonding due to its low electronegativity and the ease of losing its single valence electron And it works..
Q: How does the size of the potassium atom affect its reactivity?
A: The larger size of the potassium atom means its valence electron is further from the nucleus and experiences less attraction, making it easier to lose and resulting in higher reactivity compared to smaller alkali metals like lithium Which is the point..
Q: What happens to the electron lost by potassium during a reaction?
A: The electron lost by potassium is gained by another atom, typically a non-metal with a high electronegativity, forming an ionic bond. This electron transfer creates a stable electron configuration for both atoms involved.
Conclusion
The Lewis dot structure for potassium (K•) serves as a fundamental visual tool for understanding its chemical behavior. In real terms, the single dot representing its single valence electron highlights its high reactivity, its tendency to form ionic bonds, and its crucial role in biological systems and various industrial applications. By visualizing this simple representation, we gain a profound understanding of potassium's chemical properties and its significant contribution to various fields. The simplicity of the Lewis dot structure belies its immense power in explaining the complex world of chemical bonding and reactivity. Mastering this concept is an essential stepping stone in your journey through the fascinating world of chemistry.
And yeah — that's actually more nuanced than it sounds.